Translate this page into:
Electroencephalogram and pitfalls in children: Benign variants and how they can be misinterpreted
-
Received: ,
Accepted: ,
How to cite this article: Shahid A. Electroencephalogram and pitfalls in children: Benign variants and how they can be misinterpreted. Karnataka Paediatr J 2021;36(1):19-26.
Abstract
Electroencephalogram (EEG) has been a useful tool in the diagnosis of epileptic and non-epileptic findings for about 100 years now. The interpretation of this test requires added training for neurologists in epilepsy and EEG reading. There are many findings that may be misread or overcalled as abnormal when the findings are actually normal variants in specific age groups. A misdiagnosis may result in unnecessary medications or other treatments that can potentially result in morbidity and even mortality, the latter being seen in incorrect diagnosis of status epilepticus leading to aggressive therapies. Available history and careful analysis of the EEG used together at the time of interpretation can avoid these sources of morbidity and mortality as well as a misdiagnosis of epilepsy.
Keywords
Pediatric EEG
EEG misinterpretation
Benign variants
INTRODUCTION
Electroencephalography (EEG) was first described by Hans Berger in 1929. Since that first description, EEG has been widely used to aid the clinician in an accurate diagnosis and management of epilepsies. While the EEG is an extremely useful tool in diagnosing epilepsy, this test should be used in conjunction with the available clinical history and any imaging studies and not as a diagnostic tool by itself.
The interpretation of the EEG should be done in a step wise and organized fashion to avoid over calling, or even under calling the findings while keeping in mind that the interpretation depends heavily on the training and the comfort level of the reader.
Understanding of normal pitfalls, variations in normal EEG and study of benign variants are important things to learn in EEG.[1] Determination of the clinical significance of focal or generalized spikes or epileptiform discharges may also be a source of confusion and a potential pitfall.[2] Therefore, a sound understanding of the EEG as a whole – one that includes normal age-specific findings, normal variants and abnormal epileptiform and non-epileptiform discharges are needed for an accurate diagnosis.
Pediatric EEGs differ from adult EEGs in many aspects. Interpretation of neonatal EEGs specifically require special emphasis and time during fellowship training. Therefore, ideally pediatric and neonatal EEGs should be read by a pediatric neurologist with additional training in epilepsy and neurophysiology.
INTERPRETATION OF THE EEG
Interpretation starts with an understanding of normal findings for different age groups in children. The neonates, for example, have very specific findings that evolve from week to week as the child grows. This is in contrast to an older child whose EEG may not change significantly as the child grows with a few minor exceptions. This is in addition to an understanding of age-specific physiologic and non-physiologic artifacts and variants that may not represent brain pathology. As the reader becomes more comfortable with accurate recognition of the normal EEG and its variants, then a study of the abnormal EEG should be undertaken.
AWAKE EEG
Recognition of a normal awake record starts with ensuring a correct montage, sensitivity, and filter settings. This is followed by confirming the presence of the awake state by noting the presence of eye movements, eye blinks, muscle artifact, and the presence of a posterior dominant rhythm in the correct range depending on the age of the child. Slight variations between the right and the left side in the amplitude of the posterior dominant rhythm may be seen and often fall within the normal range of acceptable variability. The reactivity of the posterior dominant rhythm to eye opening or closure clearly distinguishes this rhythm from any possible pathological rhythms. Posterior dominant rhythm that is reactive to eye opening appears between 3 and 4 months of age in about 75% of full-term infants.[3]
The response of the normal brain to activating procedures such as hyperventilation and photic stimulation should also be carefully analyzed as these may sometimes be confused with abnormal brain activity.
NORMAL VARIANTS IN AWAKE EEG
In addition to findings seen consistently in all the patients such as eye blinks and posterior dominant rhythm, there are other findings or variants that may be seen in certain circumstances or in a small group of patients. Understanding the nature, location, morphology, and setting of the variants is essential to not confuse them with abnormal discharges or as abnormal findings.
A common occurrence, especially in active children with difficult set ups, is an inaccurate electrode placement, such that the distances between the electrodes are either too short or too large. This may misguide the interpreter to believe that there is an asymmetry of voltage when in fact the electrode placement is inaccurate.
Mild voltage asymmetries in the posterior dominant rhythm between the right and the left hemispheres are common and should not be overcalled. In almost all the children, an asymmetry between the two hemispheres of about 20% may be seen with lower voltage seen on the left side.[3]
Mu rhythm is a normal rhythm in the alpha range located in the central regions. Only about 5% of children under the age of 4 years would have a clearly defined mu rhythm. The incidence then increases with age until it reaches 18% by 16 years of age. This rhythm is usually seen more often in girls than boys.[4,5] This rhythm has an arch like morphology [Figure 1] and does not block with eye opening, but does block with movement of the contralateral extremity.
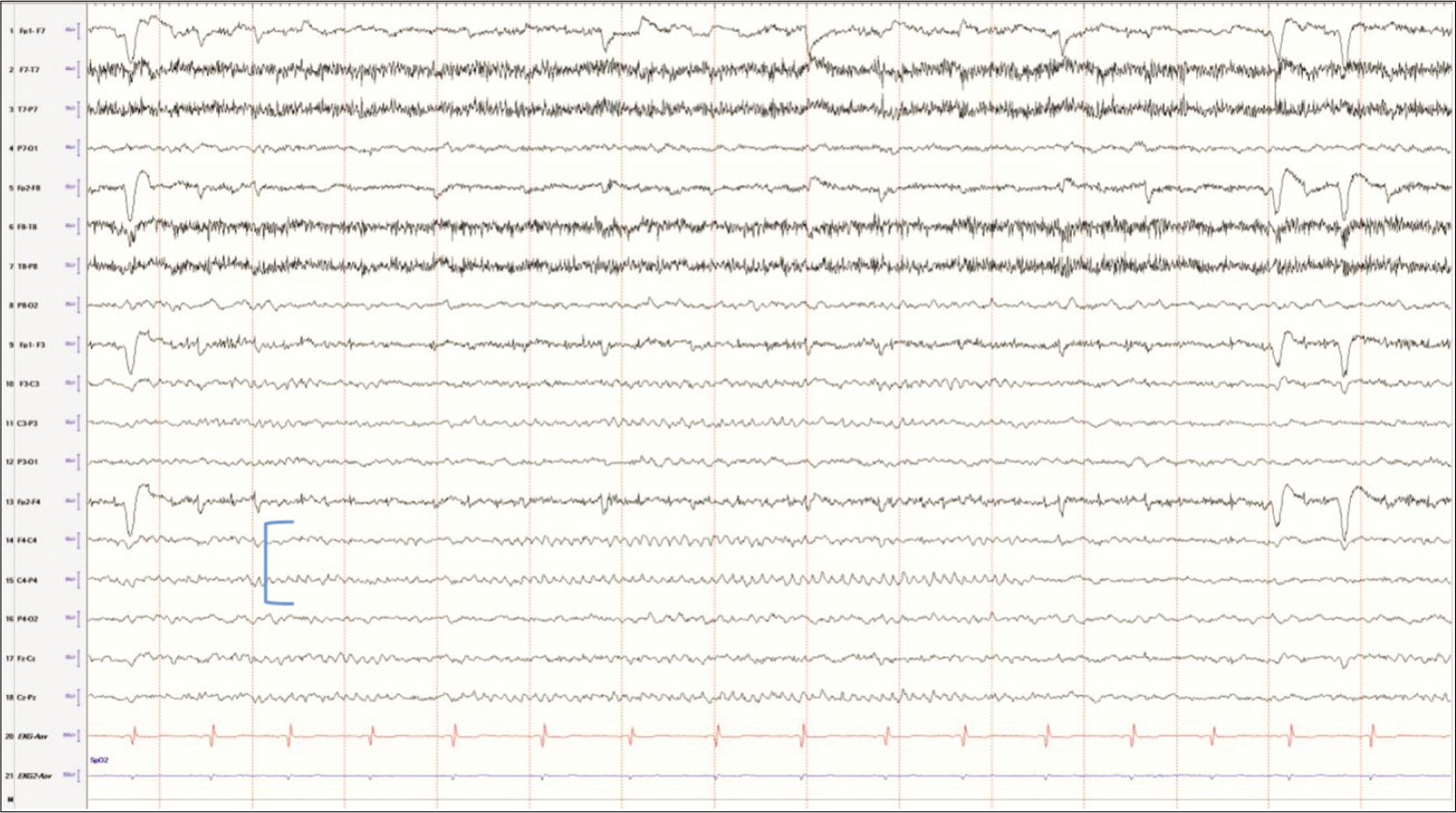
- Mu rhythm in a developmentally normal 9-year female.
Mu rhythm is usually seen only on one side at a time and the voltage is prominent and higher than the rest of the EEG.[6] This rhythm is not consistently seen and may be easily confused with asymmetry, especially due to a skull defect as may be seen with bur hole or with prior history of brain surgery. This abnormal asymmetry would be a continuous finding and may be associated with underlying slowing, while mu rhythm would not be associated with underlying slowing and is not continuous [Figure 2].
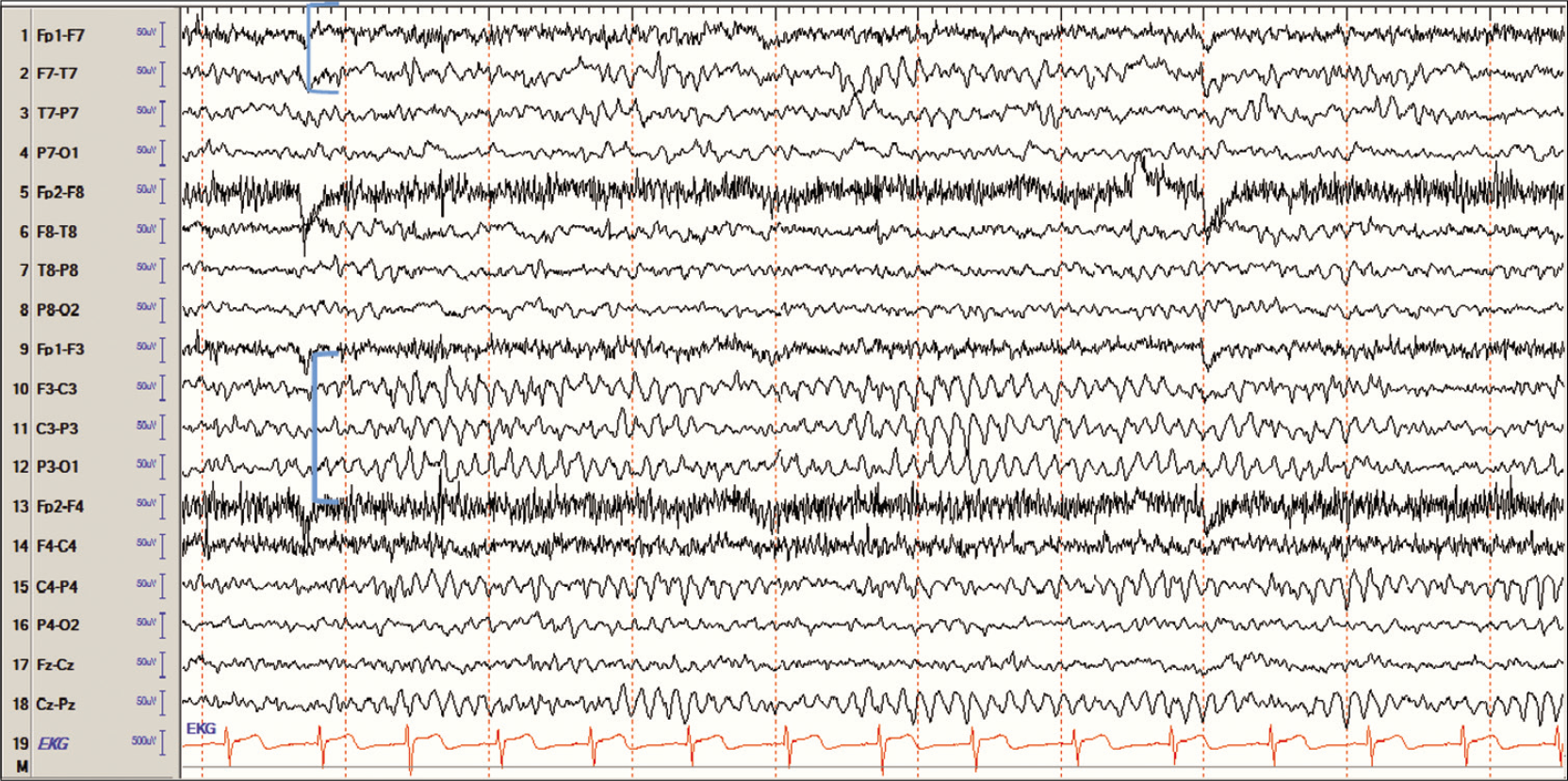
- Asymmetry due to skull defect (breach rhythm) in the left central region in an 11-year-old girl with a history of epilepsy surgery. Note the slowing in the left temporal chains indicating cortical dysfunction – in this case, the resection.
Lambda waves may be recorded bilaterally in the occipital regions in a child who is alert and awake with eyes open and likely scanning a book or a complex picture. They are surface positive, however in a child, these waves could have a sharp component that is surface negative. They may be asymmetrically present only on one side.[3] The presence of these waves only on one side can lead the interpreter to believe the waveforms to be pathologic spikes. Young children can have benign focal epilepsies of childhood originating in the occipital regions, and one is certainly prone to mistakenly identify this normal variant as an abnormal finding. One can differentiate between the two by asking the child to close their eyes. Lambda waves would disappear with eye closure, while occipital spikes would persist [Figures 3 and 4].
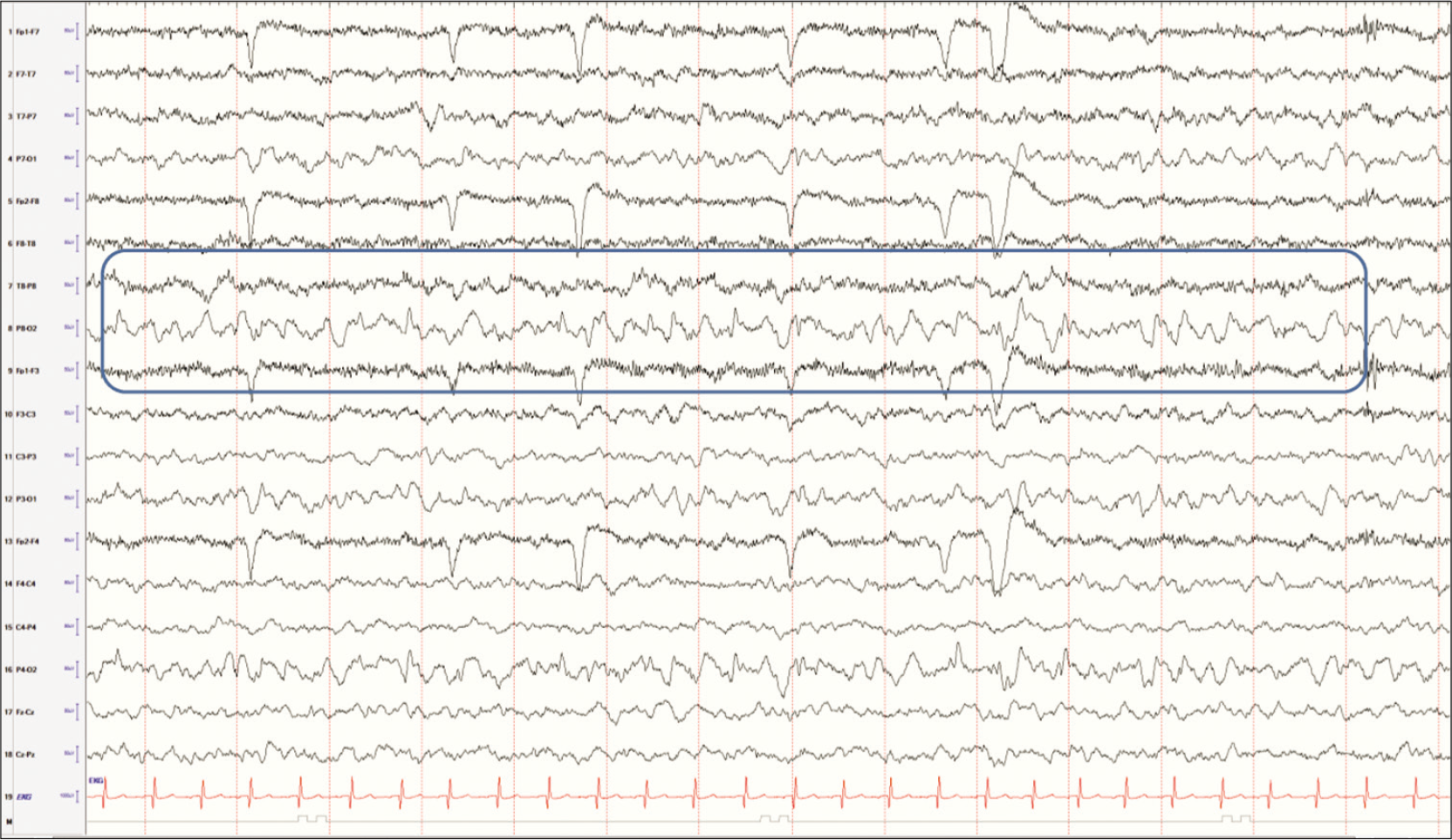
- Posteriorly located biphasic sharp waves in an awake child scanning a complex picture consistent with lambda waves.
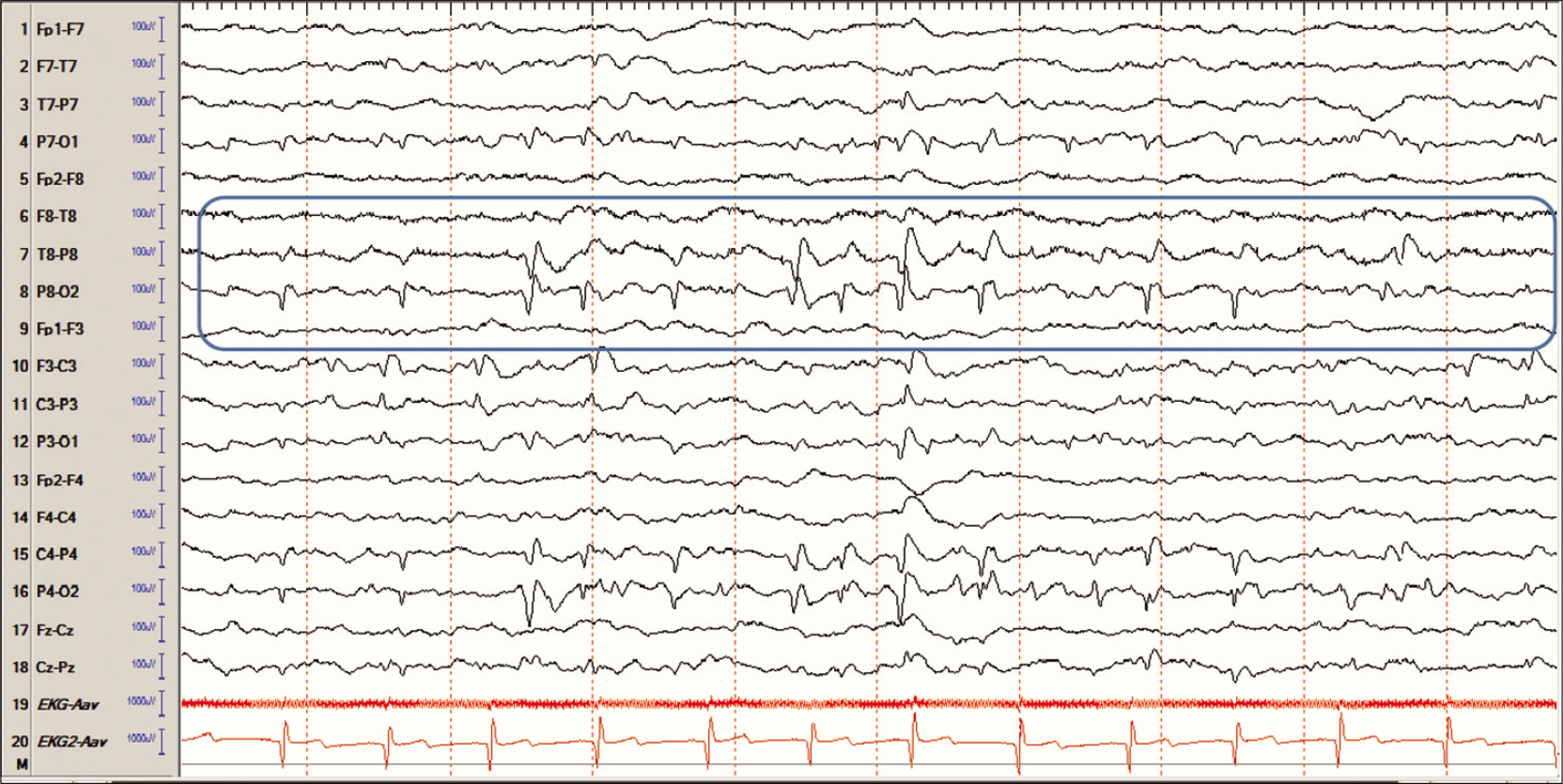
- Occipital spikes with a prominent negative component and a field extending anteriorly in a child who is sleeping. Compare the morphology and the state of the patient with lambda waves.
Hyperventilation, an “activation procedure” is a standard part of any routine or long-term EEG in a cooperative patient. The patient is asked to hyperventilate for 3–5 min and response on the EEG is noted. Most common response seen in children is a gradual buildup of delta frequencies with voltages reaching up to 300 microvolts. This activity may be more prominent in the occipital regions and may persist for about a minute or so after cessation of the activation procedure. More robust responses are seen in children between the ages of 8 and 12 years.[3,7] This high voltage, paroxysmal rhythmic activity may sometimes be confused with sharp waves or encephalopathy. While 3 Hz spike-and-wave discharges may be seen in children with absence epilepsy, the organization and abrupt onset and offset of the spike-and-wave complexes make it easier to differentiate from physiologic hyperventilation activity.
SLEEP EEG
Sleep EEG findings start when the child shuts their eyes clinically with corresponding changes in the EEG that includes fragmentation or disappearance of the posterior dominant rhythm, and overall slowing of the EEG with a loss of the anterior to posterior gradient. There is also disappearance of eye blinks and muscle artifact with an appearance of slowing roving eye movements and possible vertex waves in the drowsy state. These vertex waves become clearer as the child continues to sleep. As the child transitions to stage 2 sleep, in addition to vertex waves, sleep spindles appear in the para sagittal regions in a synchronous fashion. However, the sleep spindles may be asynchronous in a young child with incomplete myelination of the corpus callosum as would be the case in a child under the age of 2 years. Slow wave sleep is characterized by generalized diffuse slowing and rapid eye movement (REM) sleep with bursts of saccadic eye movements with a lack of eye blinks and muscle artifact.
NORMAL VARIANTS IN SLEEP EEG
The most common mistake by an interpreter not used to the pediatric EEGs is the presence of vertex waves [Figures 5 and 6]. In younger children, these waves can have a sharper appearance than older children and adults. These can also appear in short bursts, especially as the child is in the early stages of sleep. This can understandably lead to an incorrect diagnosis of epilepsy if attention is not given to the state change, the morphology of the waves and the distribution of the waves. In terms of distribution, vertex waves would be expected to be seen predominantly in the parasagittal regions and decrease in frequency or disappear as the child drifts into stage 2 or stage 3 sleep.
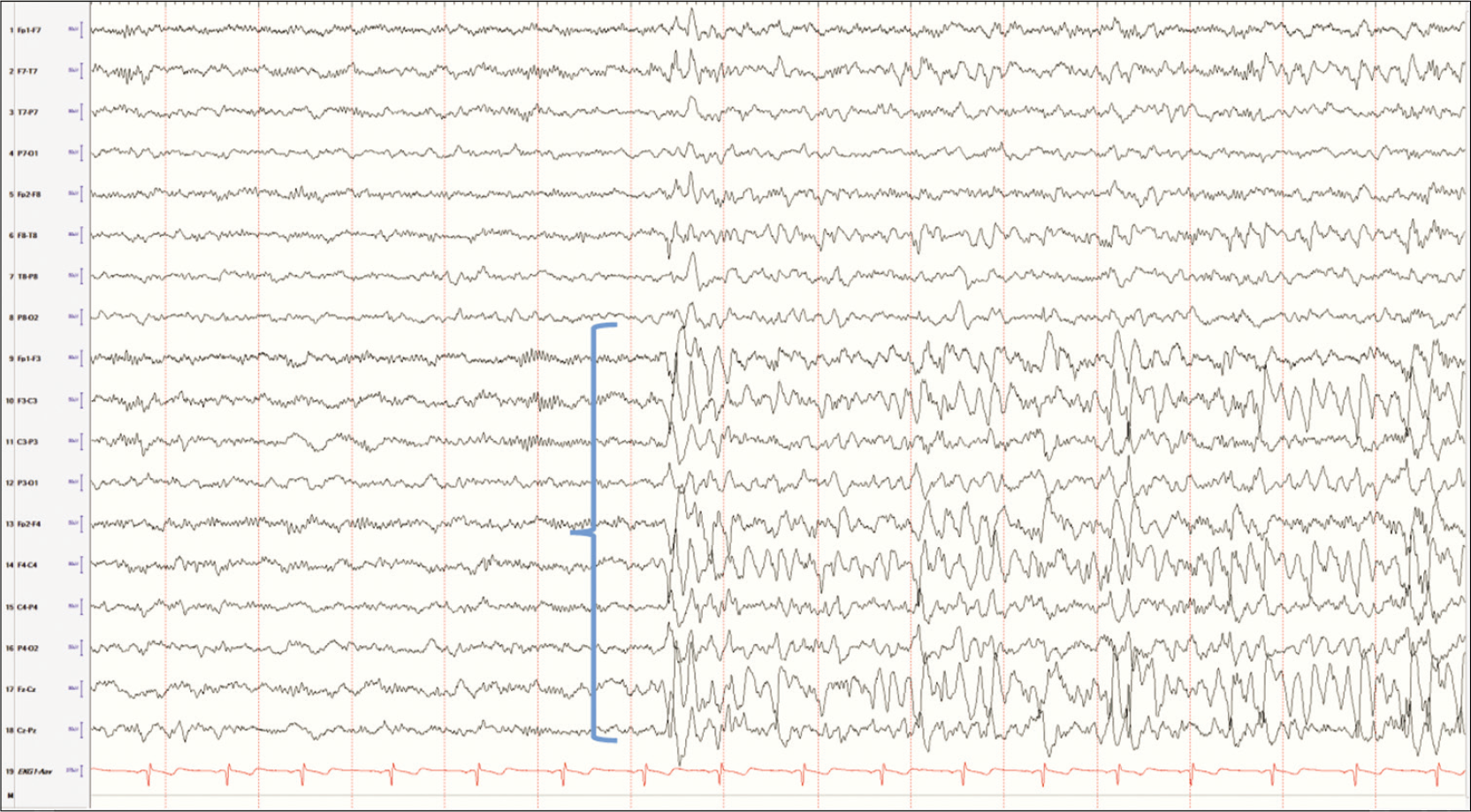
- Vertex limited to the parasagittal electrodes in early stages of sleep in a young child.
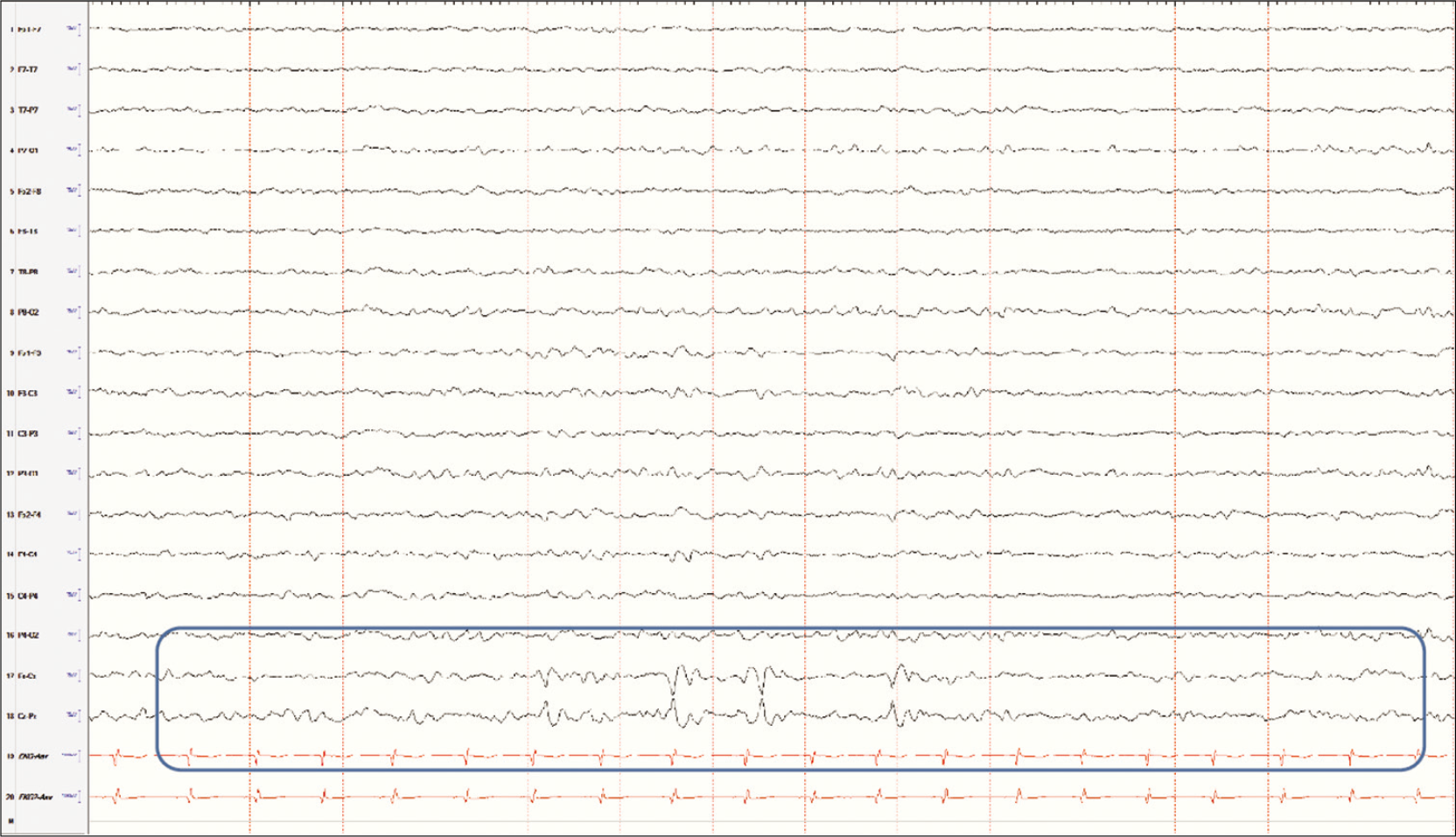
- Vertex waves limited to the Cz electrode in sleep.
It is important to note that vertex waves are a normal phenomenon related to early sleep stages and should not be confused with epilepsy arising from the vertex or the parasagittal regions. In the case of focal epilepsy, the spikes would be present in the awake state in addition to sleep, may have an after-going slow wave and persist through different sleep stages and may also be associated with focal slowing in the affected region [Figure 7]. This finding then should be taken in the context of the seizure semiology to arrive at the correct diagnosis.
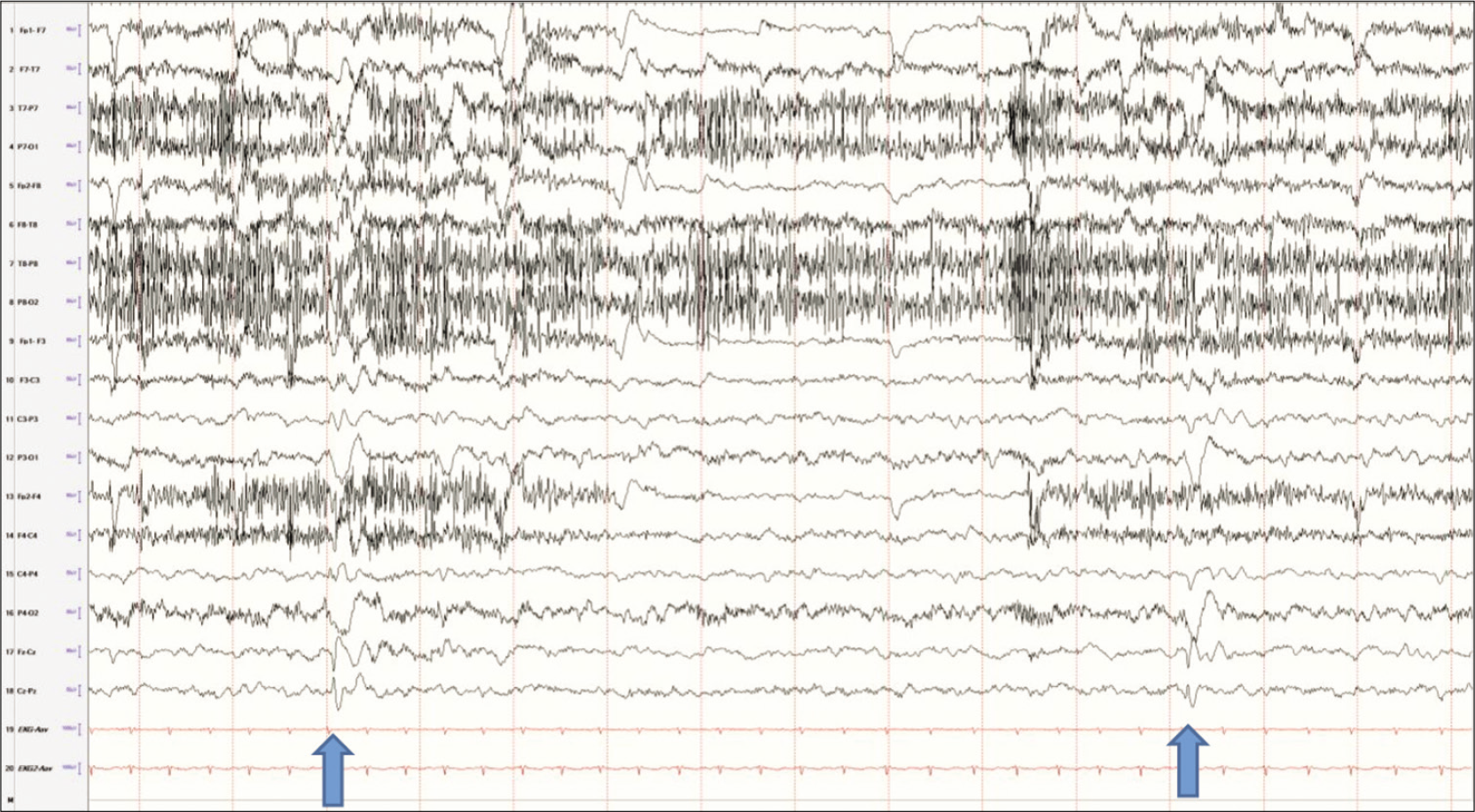
- Spikes in the vertex electrodes, maximum in Cz (arrows). Note the difference between these waves and the vertex waves. These are present while the patient is awake, which vertex waves would not be seen in wakefulness.
A child with lambda waves in wakefulness may also display positive occipital sharp transients of sleep or POSTS. A study by Egawa et al. reported the highest incidence of POSTS in the first 30 min of non-REM sleep after falling asleep.[8] This is a common finding mostly seen in older children. Again understanding the principles behind “distribution” of waveforms become essential to characterize these waveforms as normal variants rather than an abnormal finding. The main characteristic that sets these waveforms distinct from epileptiform discharges is that POSTS are surface positive, as opposed to spikes or epileptiform discharges which are surface negative. These abnormal waveforms may be present through different stages of wakefulness and sleep. A closer analysis of the morphology of the waveforms would also reveal a clear difference from epileptiform spikes, in addition to a slow wave and the absence of slowing in the background [Figure 8].
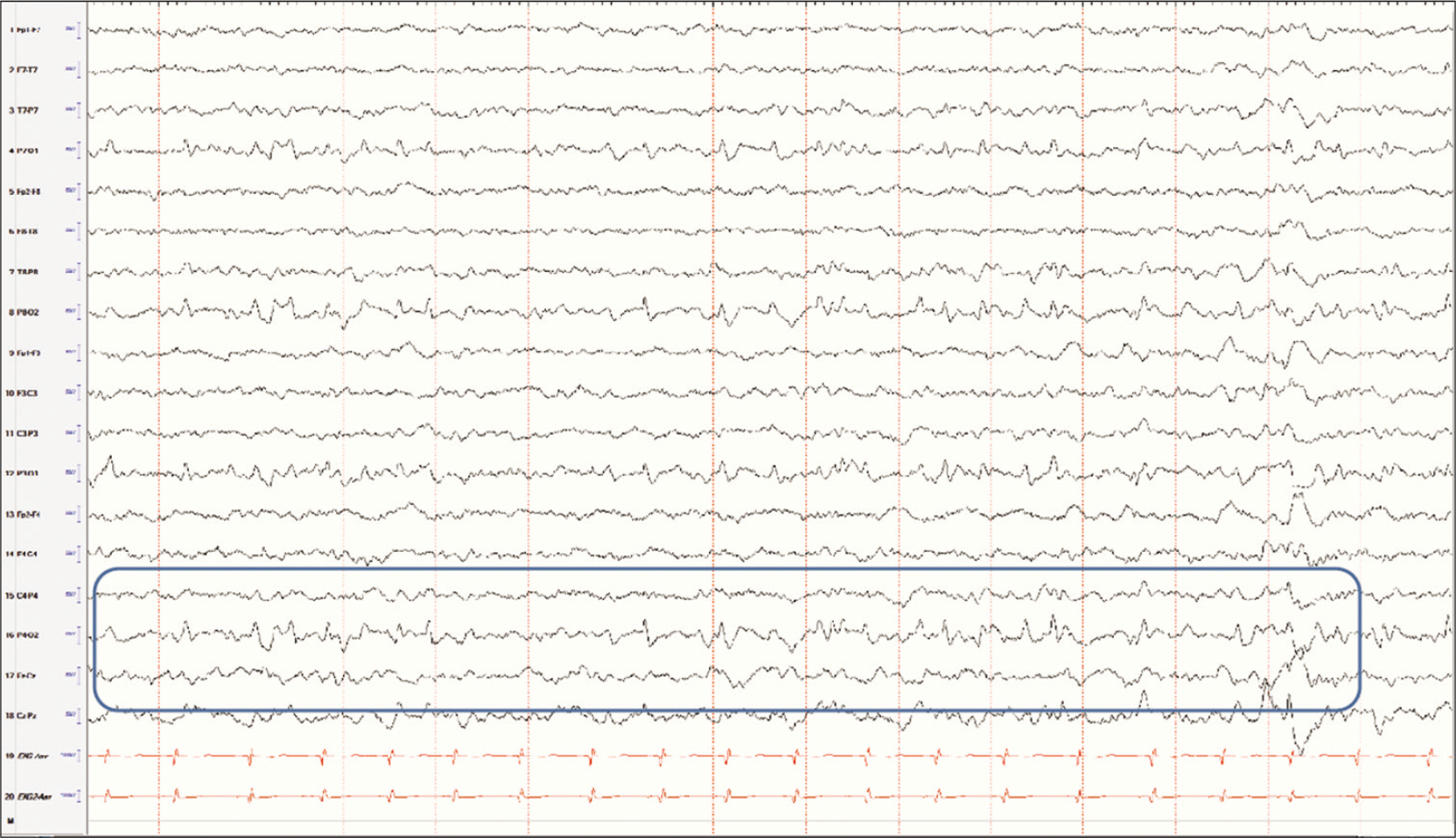
- POSTS seen in a child with no history of epilepsy in sleep. The morphology of these waveforms resemble those of another benign variant, the lambda waves and is not to be confused with occipital spikes.
Another normal pattern that can cause confusion is the 14 Hz and 6 Hz positive bursts. These are commonly seen in drowsiness and light sleep stages. The faster frequency is more common than the slower one. These waveforms appear in short bursts lasting about 1 s in duration and have a positive “spikey” appearance and the negative wave is rounded.[3] The location of these discharges is the posterior temporal region [Figure 9]. Given the spikey appearance, these waveforms may be confused with polyspikes. When analyzed closely, one would see that the spikey component is actually of positive polarity, as opposed to an epileptiform discharge which would have a negative polarity for that component of the waveform. The other clue that separates the 14 and 6 positive bursts from pathologic polyspikes is the arch-like morphology, the absence of after-going slow wave, and the absence of slowing of the background at the time of the appearance of these bursts. Another clue is the appearance only in early sleep stages and their absence in the awake state.
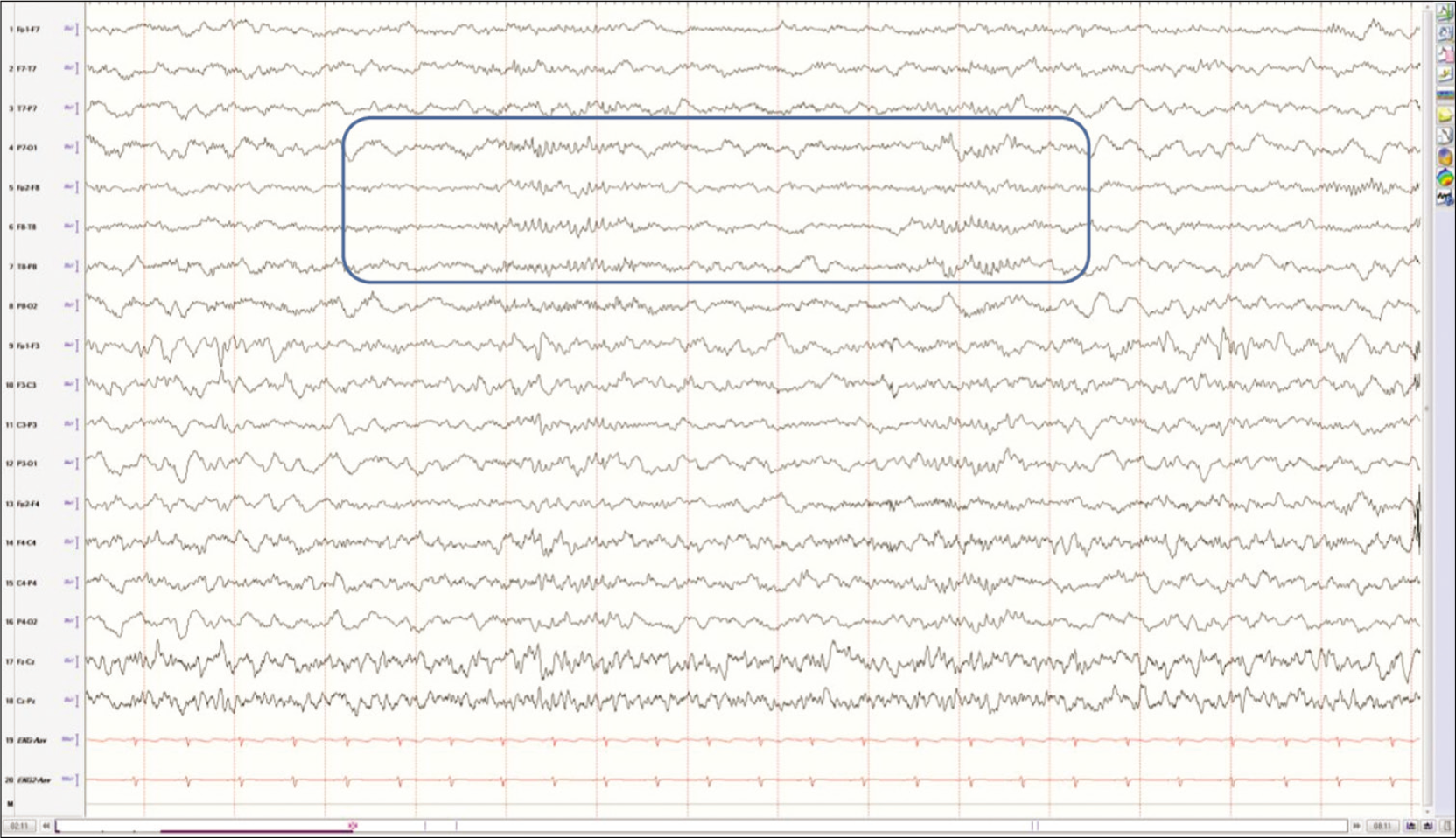
- Fourteen and six positive bursts in an 8-year-old boy in light sleep stages. The bursts are seen in both hemispheres and last about 1 s in duration.
WHERE DOES THE INTERPRETATION GO WRONG?
The key to not over calling findings on the EEG is related to an understanding of the basis of a seizure and interictal epileptiform discharges. This is in addition to an understanding of the maturation of the brain for a pediatric neurologist/epileptologist. The former applies to a pediatric reader and the latter to someone who is interpreting neonatal EEGs. Another important aspect is to understand the “art” of analyzing a waveform in depth rather than simple pattern recognition. The analysis would help with understanding the true polarity of the waveform, or a segment of a waveform (the spikey component), and will help in determining the field of the waveform in question. Another aspect to remember is that normal variants would have a broader field on the EEG than epileptiform discharges, and shifting lateralization between the left and the right sides.[9]
Once the analysis has been completed, the reader then should be able to conclude what the true nature of a waveform truly is.
CONCLUSION
An EEG is an extremely useful tool in the diagnosis of seizures and even specific epileptic syndromes. However, its utility is a lot broader than just epilepsy and helps greatly in the diagnosis of non-epileptic events, delineation of the degree of encephalopathy, and plays a role in assisting the clinician in the prognosis.
The interpretation of this test greatly depends on the reader. The interpretation is especially challenging in the pediatric patients given the vast majority of variants seen in this age group as well as the artifacts related to movement in young children. A correct interpretation, therefore, requires the reader to have experience in reading pediatric EEGs and to be comfortable with recognition of normal variants and be able to separate these from pathologic findings. When in doubt, the electroencephalographer should look at the bigger picture and incorporate clinical history, imaging studies, and a careful analysis of the findings in question.[10] Benbadis and Tatum in their paper reported that most of the overcalled patterns happened to be normal fluctuations of the background.[11] Overcalling the findings may result in unnecessary medication use and wrong diagnoses.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Avoiding the pitfalls of EEG interpretation in childhood epilepsy. Epilepsia. 1996;37(Suppl 1):S41-51.
- [CrossRef] [PubMed] [Google Scholar]
- The development of the electroencephalogram in normal adolescents from the age of 16 through 21 years. Neuropadiatrie. 1971;3:11-45.
- [CrossRef] [PubMed] [Google Scholar]
- The development of the electroencephalogram in normal children from the age of 1 through 15 years. Non-paroxysmal activity. Neuropadiatrie. 1971;2:247-304.
- [CrossRef] [PubMed] [Google Scholar]
- Handbook of Electroencephalography (1st ed). Bogota, Colombia: Universidad de Los Andes; 2013.
- [Google Scholar]
- Positive occipital sharp transients in the human sleep EEG. Folia Psychiatr Neurol Jpn. 1983;37:57-65.
- [CrossRef] [PubMed] [Google Scholar]
- Normal variants are commonly overread as interictal epileptiform abnormalities. J Clin Neurophysiol. 2019;36:257-63.
- [CrossRef] [PubMed] [Google Scholar]
- Interictal EEG and the diagnosis of epilepsy. Epilepsia. 2006;47(Suppl 1):14-22.
- [CrossRef] [PubMed] [Google Scholar]
- Overintepretation of EEGs and misdiagnosis of epilepsy. J Clin Neurophysiol. 2003;20:42-4.
- [CrossRef] [PubMed] [Google Scholar]






