Translate this page into:
Genesis and evolution of KIMIE: New cost-effective indigenous human breast milk pasteurizer
*Corresponding author: Shilpa Kalane, Department of Neonatology, Deenanath Mangeshkar Hospital, Pune, Maharashtra, India. drshilpakalane@icloud.com
-
Received: ,
Accepted: ,
How to cite this article: Waghmare S, Kharche A, Kalane S, Haridas V, Devaskar U. Genesis and evolution of KIMIE: New cost-effective indigenous human breast milk pasteurizer. Karnataka Paediatr J 2021;36(2):64-8.
Abstract
Natural mother’s breast milk is the best food for all newborns, especially preterm babies. However, if mother’s milk is unavailable or donor breast milk (DBM) is inadequate, DBM is the next best choice. Human milk pasteurizers are traditionally big, costly, and require special electrical and water connections, as well as need a large volume of water that is not recycled and an ongoing supply of disposable plastic bottles. Operation of these machines necessitates specialized training. The creation of Kimie, a compact, automated, and user-friendly human breast milk pasteurizer capable of pasteurizing small volumes of DBM, is described here. This system needs no special water plumbing, recycles water, and is reasonably priced.
Keywords
Human milk bank
Pasteurizer
Breast milk
Neonate
INTRODUCTION
Pasteurized donor breast milk (PDBM) is the next best feeding option for neonates when natural mother’s breast milk (BM) is not available, insufficient, or contraindicated.[1,2]
GENESIS
A female baby (nick name KIMIE), B.W. 850 g, G.A. 27 weeks, was born to G4 P3 mother by vaginal delivery. By the 3rd day, while treated with moderate respiratory support and 30 ml/kg/day of expressed breast milk (EBM), the mother died unexpectedly. Since the other siblings were also premature, baby’s father was familiar with prematurity related morbidities including NEC. He expressed a strong desire not to feed any formula. Various feeding options were discussed in detail. The level II NICU at Centinela Hospital (CH) did not have the tissue license (State of California) to use DBM obtained from a donor breast milk bank (DBMB). One nurse suggested of using another mother’s EBM who had a premature baby born at 32 weeks GA. This lady was negative times two for HIV, RPR, and hepatitis B and was rubella immune. Her EBM was in excess, well tolerated by her son, and clinically, there was no suspicion for bacterial contamination. Although there was difference of opinion, there was consensus that the risk of developing NEC due to the use of formula was high as compared to the potential of acquiring bacterial or viral infection using raw donor breast milk (DBM). While use of unpasteurized DBM may be an accepted practice in other countries, there was no such precedence in the health-care system. There was a discussion among the NICU team, the hospital administrator, father of the baby, and the potential BM donor. After extensive discussion and obtaining informed written consent by everyone, decision was made to use unpasteurized DBM milk.
After extensive search regarding availability of small breast milk pasteurizer (BMP), SafeGard used in the dairy industry was discovered [Figure 1].

- SafeGard used in dairy industry.
An extensive search regarding the availability of a small BMP was performed. SafeGard used in the dairy industry was discovered [Figure 1]. While this pasteurizer uses the principle of holder pasteurization, cooling is performed manually using ice-cold water [Figure 2]. For the next 5 days, DBM pasteurized using SafeGard was fed. Subsequently, DBM obtained from the commercial DBMB was used till the baby was discharged.
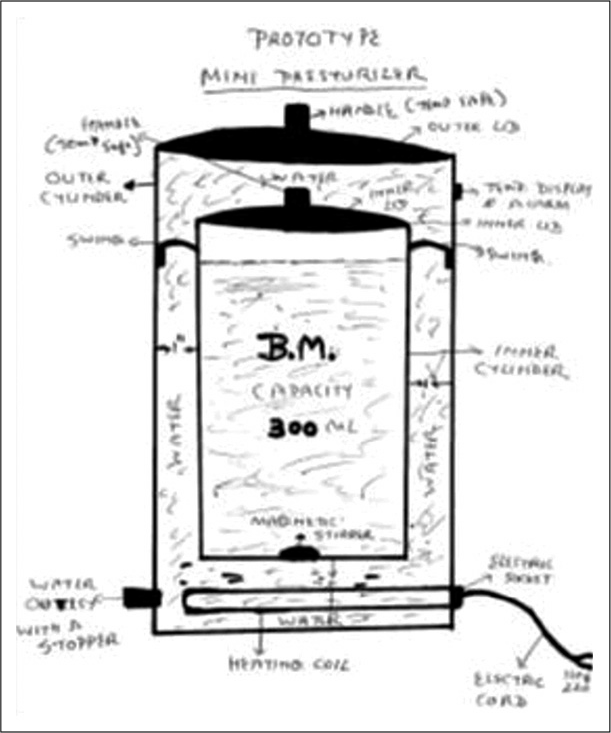
- Prototype mini-pasteurizer.
Based on the basic design of the SafeGard, modified prototype was developed by Dr. Devaskar [Figure 2]. The concept was discussed with the chief of the biomedical engineering department at UCLA. While the idea to develop first prototype was introduced, it needed two engineers for at least 6 months needing initial investment of ~500,000 dollars was anticipated.
Thereafter, UD, VH, and SUK met with two engineers SW and AK. It was concluded to develop a portable human BMP capable of pasteurizing 300 ml during each cycle using the holder technique incorporating modern automated technology for heating, holding, and cooling [Figure 3].
- Holder technique.
EVOLUTION
It was discovered that within the existing pasteurizers used in human milk banks (e.g. Sterifeed, HSC, etc.), the plumbing is connected to a water supply for filling the chamber and a separate plumbing is required to empty the water. A submersible heater of very high power is employed. To distribute heat equally, they incorporate a water stirrer. Use of a submersible water heater was concerning due to a risk of electrical shock. In addition, the waste of enormous quantity, ~80 L, of water once every cycle was problematic. Numerous choices to manufacture a compact, portable, user- and eco-friendly, and cost-efficient device using modern technology were thought of. It was determined to use two separate water tanks, one for heating and one for cooling. Two totally different water pumps were connected to the respective tanks. During this closed-loop system, every pump propels water within the chamber that warms or cools DBM and water returns to its respective tank. The capability of heating and cooling tanks was arbitrarily decided to be 1750 and 2750 ml, respectively. Thus, only 4250 ml of water would be used and will be recycled. Other discussions included the technical and tubing design, the method of water circulate across the milk container, the components required and their placement, the kind of water pumps for heating and cooling, the specified flow rate, its sturdiness, the kind of heaters and their wattage, the sort of cooling mechanism (chiller) and their ability, the scale, and form of DBM container. The first prototype capable of pasteurizing 300 ml of EBM during each cycle was developed within 3 months [Figure 4]. Then, the second prototype was developed [Figure 5]. The major limitation of the prototypes was: They could pasteurize a single sample of 300 ml during each cycle. Finally, the third prototype able to pasteurizing five different EBM samples and a total of 500 ml were developed [Figure 6]. The information of the engineering and bacteriologic sterility information has been described [Table 1]. In short, to be eco-friendly, for easy cleaning and to be cost-effective, grade 304 stainless steel cylinders rather than the disposable plastic bottles were used. In this prototype, Perplex that’s a better cloth as compared to plastic is used.

- Kimie 300 – first prototype.

- Kimie 500 – second prototype.

- Kimie 500 – third prototype.
| Characteristic | KIMIE | Sterifeed | HSC | |
|---|---|---|---|---|
| 1. | Method of Pasteurisation | Holder | Holder | Holder |
| 2. | Space Needed | 10 sq ft | 80 sq ft | 120 sq ft |
| 3. | Portability | Yes | No | No |
| 4. | Pasteurization Capacity Minimum: Maximum: |
10 ml 500 ml |
1000 ml 9000 ml |
1000 ml 3000 ml |
| 5. | Special Plumbing | No | Yes | Yes |
| 6. | Special Electrical Connection | No | Yes | Yes |
| 7 | Electric Consumption-Watts | 750 | 2800 | > 3,500 |
| 8. | Time needed per cycle-minutes | 70 | 120 | 180 |
| 9. | Water required and recycled | 4.75 litres, yes. | 85 litres, no. | 80 litres, no . |
| 10. | Cooling time (63 to 25* C)-minutes | 2 | 10 | 10 |
| 11. | Operation | Fully Automatic | Fully Automatic | Fully Automatic |
| 12. | Cooling mechanism | Inbuilt | Not inbuilt | Not inbuilt |
| 13. | Sensor for milk temperature | Yes | Yes | Yes |
| 14. | Sensor for water temperature | Yes | Yes | Yes |
| 15. | Digital display in real time (Temperature and time) | Yes | Yes | Yes |
| 16. | Noise Level | < 50 DbA | > 50 DbA | > 50 DbA |
| 17. | Material used for the milk container and reused | Stainless steel, yes | Plastic, no | Plastic, no |
| 18. | Auto drain | Yes | Yes | Yes |
| 19. | Cycles / day | Several | Hard to use more than thrice | Hard to use more than thrice |
In 2018, Kimie obtained the ECU certificate of compliance (no 1810170911101) for a human milk pasteurizer and met standards EN 6060-1, EN 60601-1-2, and EN 60601-1-6 to illustrate conformity. In the USA, BM milk pasteurizer does not require FDA approval as it is considered a catering device. Kimie was imported to CH underneath commodity code 84198998.
Kimie [Figure 6] has been successfully used in several hospitals in India for more than 2½ years and at CH for >10 months. It was quickly recognized that there has been a need for production a system able to pasteurizing extra than 500 ml. Kimie [Figure 6] is the today’s prototype able to pasteurizing 3000 ml during each cycle and has six different cylinders like Kimie-500. Selling price of Kimie-500 and Kimie-3000 is 5 and 8 lakh rupees, respectively.
DISCUSSION
The origins and evolution of a home-grown, compact, portable, fully automated, user-friendly, cost-effective BMP that uses modern technology, needs less space, has no special plumbing, and can recycle water are described in this paper. Stainless steel is preferred by regulatory agencies in the food industry for heating, cooling, and drying food because it avoids reactions between the food and the container. Safety, durability, corrosion resistance, non-absorbency, adequate weight and thickness to withstand repeated washing, a smooth, easy-to-clean surface, and resistance to pitting, chipping, crazing, scratching, and scoring are all requirements. As a result, stainless steel grade 304 bottles are used rather than plastic bottles.
The indications for using pasteurized BM have been listed in detail and are briefly discussed below.[3]
Premature infants
There is a growing trend to begin enteral feeding within few hours after birth and progress to full feeds as quickly as possible. The best feeding choice for a baby is his or her mother’s EBM, which includes colostrum. Supplementing with PDBM is a safer choice when mother’s EBM milk is unavailable, inadequate, or unsuitable.[1-5] DBM from another baby’s mother can be collected, pasteurized, and frozen, or it can be pasteurized shortly after expression and used right away. This method would reduce the need for TPN and minimize the use of premature formula or DBM acquired from mothers who delivered their babies at full term.
Prevention of cytomegalovirus (CMV) transmission from the use of the natural mother’s BM
A recent study reported an association between postnatal CMV infection and negative outcomes.[6] Pasteurization of BM can help to prevent CMV transmission since holder pasteurization eliminates the virus. Many times pasteurizing a baby’s own mother’s EBM is not feasible due to the lack of readily available pasteurization machines. It is easier to pasteurize the natural mother’s BM to remove CMV now that Kimie is available. We have enrolled five babies born at <32 weeks of GA who have received own mother’s EBM pasteurized using Kimie.
Prevention of CMV and HIV-1 transmission
Feeding accounts for half of mother-to-child transmission of HIV-1.[6,7] Coinfection with CMV in the BM is linked to an increased risk of HIV-1 transmission from mother to child after birth. In countries with limited resources, BM is still critical for infant survival. As a result, eliminating CMV and HIV-1 virus by pasteurizing mother’s BM before ingestion will prevent vertical CMV and HIV-1 transmission. It should be possible to do so now that Kimie is available.
Prevention of CMV and other viral infection in immune-compromised infants
Severe combined immunodeficiency (SCID), 22q11 deletion syndrome (DiGeorge), CHARGE syndrome, VACTERL association, and ataxia telangiectasia are a few examples of primary immunodeficiencies.[6-11] It is not recommended to breastfeed or use PDBM to avoid viral transmission.[6-11] One baby was diagnosed with SCID at CH at the age of 9 days. He has been getting his own mother’s BM, pasteurized with Kimie, for the past 10 months. Although awaiting the HSCT, he has remained CMV negative.
Prevention of COVID-19 transmission
In general, if the mother is COVID-19 positive at the time of birth, her baby will unlikely be colonized by the virus. Similarly, her BM tests negative for the virus at birth. However, if these women do not take the necessary precautions, the COVID-19 virus may infect their BM. Pasteurization of her EBM may be indicated in these circumstances, especially in the preterm baby. Holder pasteurization of milk has been shown to eliminate the COVID-19 virus in a recent study.[12]
CONCLUSION
Necessity breeds ingenuity, and disaster breeds opportunity. Kimie’s development and evolution is an excellent example of clinicians and engineers collaborating. More partnerships like this are needed with the common aim of improving the health of neonates all over the world.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Human milk-treatment and quality of banked human milk. Clin Perinatol. 2017;44:95-119.
- [CrossRef] [PubMed] [Google Scholar]
- Formula versus donor breast milk for feeding preterm or low birth weight infants. Cochrane Database Syst Rev. 2014;4:CD002971.
- [CrossRef] [PubMed] [Google Scholar]
- Breastfeeding and the use of human milk. Paediatrics. 2012;129:e827-41.
- [CrossRef] [PubMed] [Google Scholar]
- Donor human milk for preterm infants: Current evidence and research directions. J Pediatr Gastroenterol Nutr. 2013;57:535-42.
- [CrossRef] [PubMed] [Google Scholar]
- Enteral nutrient supply for preterm infants: Commentary from the European society of paediatric gastroenterology, hepatology and nutrition committee on nutrition. J Pediatr Gastroenterol Nutr. 2010;50:85-91.
- [CrossRef] [PubMed] [Google Scholar]
- Human breast milk-acquired cytomegalovirus infection: Certainties, doubts and perspectives. Curr Pediatr Rev. 2019;15:30-41.
- [CrossRef] [PubMed] [Google Scholar]
- Inactivation of human immunodeficiency virus Type I in human milk: Effects of intrinsic factors in human milk•and of pasteurization. J Hum Lact. 1993;9:13-7.
- [CrossRef] [PubMed] [Google Scholar]
- Inactivation of high-risk human papillomaviruses by Holder pasteurization: Implications for donor human milk banking. J Perinat Med. 2014;42:1-8.
- [CrossRef] [PubMed] [Google Scholar]
- Short communication: Pasteurization of milk abolishes bovine herpes virus infectivity. J Dairy Sci. 2005;88:3079-83.
- [CrossRef] [Google Scholar]
- Inactivation of lymphocyte-transforming activity of human T-cell leukemia virus Type I by heat. Jpn J Cancer Res. 1986;77:13-5.
- [Google Scholar]
- Hepatitis B viral markers in banked human milk before and after Holder pasteurization. J Clin Virol. 2009;45:281-4.
- [CrossRef] [PubMed] [Google Scholar]
- Holder pasteurization of donated human milk is effective in inactivating SARS-CoV-2. CMAJ. 2020;192:E871-4.
- [CrossRef] [PubMed] [Google Scholar]






