Translate this page into:
Skin prick testing in children
*Corresponding author: Srinivas Jakka, Consultant in Paediatrics, Pulmonology and Allergy, Ankura Hospitals, Kukatpally, Hitech City Road, Hyderabad-500072, Telangana, India. jakkasrinu@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Jakka S. Skin prick testing in children. Karnataka Paediatr J 2020;35(2):67-71.
Abstract
There is an increasing incidence of allergies across all the ages in India. Pediatricians of the future will increasingly be encountering various allergic conditions. Accurate diagnosis of allergies depends very much on a detailed history supported by the judicious use of allergy tests. The two widely available allergic tests in our country are the skin prick testing (SPT) and serum-specific IgE. This article discusses in detail about the various aspects of SPT including the indications, technique of SPT, interpretation of the results, advantages, and limitations of SPT.
Keywords
Skin prick testing in children
Diagnosis of allergy in children
Allergy testing in children
INTRODUCTION
There is an increasing incidence of allergies across all the ages in India.[1] Pediatricians of the future will increasingly be encountering various allergic conditions such as atopic dermatitis, food allergies, allergic rhinitis, and asthma in their day-to-day practice. Accurate diagnosis of allergies depends very much on a detailed history supported by the judicious use of allergy tests. The history should focus first on confirming the diagnosis of allergy and then identifying the potential allergen.[2] Investigations should be guided by the history and performed judiciously. Blanket testing will only lead to confusion to both the patient and the doctor. Allergic reactions can be IgE mediated, non-IgE mediated, or a mixture of both. This understanding is important because the commonly used allergy tests diagnose only the IgE-mediated reactions. It is also important to differentiate between sensitization and clinical allergy. A person is said to be sensitized when he makes IgE antibodies against the allergen (demonstrated by a positive skin prick testing [SPT] or specific IgE [sIgE]). However, he may or may not have clinical allergy (demonstrated by symptoms when exposed to the allergen). This knowledge is important when interpreting the allergy tests. The two widely available allergic tests in our country are the SPT and serum sIgE. SPT is considered the gold standard for aeroallergens,[3] while double-blinded placebo-controlled food challenge remains the gold standard for food allergy.[4] However, as oral food challenge is dependent on expertise and resources, physicians use SPT and sIgE for diagnosis of food allergy as well.
BLOOD TESTS
Non-specific tests such as total IgE and absolute eosinophil count are not useful in the diagnosis of allergy as these can be elevated in many other conditions.[5] The blood investigation of choice is serum sIgE to individual allergens. The standardized method for sIgE available in India is the ImmunoCAP test[6] that uses fluorescent enzyme immunoassay technique. The advantages of sIgE are the safety, availability, and reproducibility.[7] The disadvantages are the false positivity, cost involved, and delayed results. The false positivity is due to cross-reactivity among the epitopes of various allergens. Component resolved diagnosis where sIgE to specific allergenic epitopes is tested, hopes to overcome this problem in the future.
SKIN TESTS
The various skin tests include the epicutaneous or percutaneous test, intradermal test, and patch test. The intradermal tests are used in limited conditions such as allergy to drugs, vaccines, and insect venom. It requires high expertise and carries greater risk of adverse effects.[8] The patch test is used for testing delayed hypersensitivity reactions like contact dermatitis. The percutaneous skin test is widely practiced as it is easy to perform, has low risk of side effects, and gives good results when performed and interpreted accurately. It can be done by scratch, puncture, and prick techniques. Of the three techniques, percutaneous SPT is the most popular and commonly used test. This article discusses further about the SPT.
INDICATIONS FOR SPT
SPT is indicated where IgE-mediated allergic mechanism is suspected such as atopic dermatitis, allergic rhinoconjunctivitis, asthma, food allergy, drug allergy, latex allergy, urticaria, and anaphylaxis.[9] Other less common conditions include allergic bronchopulmonary aspergillosis, eosinophilic esophagitis, and gastroenteritis.
The common inhaled allergens (aeroallergens) across the world are house dust mites, pollen, animal dander, molds, and cockroaches. The common food allergens across the world are cow’s milk, hen’s egg, soy, wheat, pea nuts, tree nuts, fish, and sea food. In addition, pediatricians should also have an understanding of the locally prevalent allergens which can vary from place to place.[10,11]
MECHANISM OF SPT
SPT uses cutaneous sensitivity as a surrogate marker for sensitization in target organs such as skin, eyes, nose, lungs, and gut.[9] Whenever an allergic person is exposed to an allergen, his immune system makes IgE antibodies. These antibodies remain attached to the surface of mast cells. During SPT, when the allergen is introduced into the skin, it interacts with IgE bound to the cutaneous mast cells. Cross-linking of the immunoglobulins occurs leading to release of histamine and other chemical mediators. This produces a local allergic reaction in the form of wheal and flare. This local reaction is measured and compared with histamine taken as positive control and normal saline taken as negative control. As the epidermis is relatively free of blood vessels and pain fibers, when performed appropriately, the SPT should not cause major discomfort or bleeding.
CONTRAINDICATIONS FOR SPT
SPT is contraindicated where there is an extensive skin disease (e.g., atopic dermatitis) with very little normal skin, skin conditions such as urticaria and severe dermatographism, poor patient cooperation, and patient unable to stop drugs that can interfere with SPT.
It should be done cautiously in very young infants, pregnant women, severe asthma, history of anaphylaxis, conditions associated with elevated tryptase levels, and patient on drugs that can increase the risk of severe allergic reactions (beta-blockers, ACE inhibitors, etc.). SPT in these circumstances should be done at controlled settings by experienced practitioners.
PREPARATION FOR SPT
We should ensure that the patients do not use drugs that can interfere with the result of SPT for the duration of action of these drugs. Antihistamines suppress the histamine response for a variable period of time. In general, first-generation antihistamines can be stopped for 72 h. Second-generation antihistamines suppress histamine response for a variable length of time, up to 7 days.[12] Leukotriene receptor antagonists do not affect the skin reactivity.[12] Short-term oral corticosteroid treatment does not seem to alter the reactivity to SPT.[13] Topical steroids can affect reactivity of SPT[14] and some studies have shown that the inhibitory effect of short-term topical steroids lasts for 3 days.[15] However, other studies have shown that application of potent topical steroids can affect SPT reactivity for 3 weeks.[16] Topical calcineurin inhibitors have a variable affect. Pimecrolimus does not affect histamine testing[17] but tacrolimus seems to affect it.[18] However, animal studies have shown that tacrolimus has no effect on immediate reactions but decreased some late-phase reactions.[19] Therefore, no withdrawal is recommended to evaluate only immediate reactions, but a 4-week withdrawal may be necessary for the evaluation of late-phase reactions.
Patients are explained about the procedure and consent obtained. They should be reassured that the test will cause only mild discomfort. All materials needed for SPT are kept ready before the test. This includes standard allergen extracts, lancets for skin prick, marker pens, and tissues for wiping the skin after the procedure. Resources to manage any severe reactions, such as IM adrenaline, oxygen source, inhaled salbutamol, IV fluids, and resuscitation equipment, should be kept ready.[8] It is important that standard commercial reagents are chosen for the procedure as the quality of reagents determines the outcome of the SPT. The commercially available allergen extracts in India have good practical utility though these are not standard. Where standard commercial allergen extracts are not available, a prick-to-prick technique can be done by pricking fresh food with the lancet and then pricking the skin.[8]
DEVICES USED FOR SPT
SPT requires a sharp pointed lancet that is used to prick through the allergen extract and into the superficial layers of the skin. The lancets come in different shapes and sizes and the choice depends on comfort, cost, and availability. The lancets can be dipped in allergen extracts and then pricked into the skin; or a drop of allergen extract is placed on the skin and the lancet pricked through it.
They can be single tip, double tipped (duo tip) or have multiple tips. They also come as single separate lancets for each allergen or have multiple point lancets to test multiple allergens simultaneously [Figure 1].
![Picture depicting different lancets available for skin prick testing.[8]](/content/113/2020/35/2/img/KPJ-35-067-g001.png)
- Picture depicting different lancets available for skin prick testing.[8]
SPT PROCEDURE
The skin is cleaned with alcohol swab before the procedure and allowed to dry. The skin of forearm and back is usually used for this purpose. The forearm is less sensitive than the back.[20] When forearm is used, pricks are done 2–3 cm away from wrist and cubital fossa. The skin is then marked with numbers to identify the corresponding allergens. Care is taken to maintain a gap of 2 cm between two allergens to avoid false-positive reactions due to overlap.[20] A drop of allergen is placed adjacent to the corresponding number. The lancet is passed through the drop and the skin at an angle of approximately 45 degrees. The device is then lifted, creating a small break in the epidermis. It is estimated that only 0.3 μl of fluid is introduced into the skin.[21] Using the lancet, the allergen is driven into the superficial layers of the skin by means of a gentle prick. The excess solution is mopped off using a tissue paper.
Performed properly, SPT should not produce major discomfort or bleeding.[8] The number of allergens used for testing depends on the clinical condition and the suspected allergens. The same lancet should not be used for testing multiple allergens. The histamine is read 10–15 min later and allergens 15–20 min later. As histamine produces wheal early compared to the individual allergens, it can be pricked at the end after the individual allergens. The diameter of the wheal is measured horizontally and vertically and the mean diameter is recorded in mm.[22] Some people take the largest diameter of the wheal into consideration. The flare is measured in similar method and recorded separately. Sometimes, irregular extensions of the wheal (pseudopods) can be noted. Their significance is not known.[8] After 20 min, the reaction may fade away and the test might have to be repeated again if not interpreted in time.
In high-risk cases, patient is observed for 20–30 min in the clinic after completion of the test.[23] The patient is observed for any signs and symptoms of systemic allergic reactions mentioned below.
The reagents should be stored in the refrigerator at the designated temperature. Care should be taken to leave the reagent outside the refrigerator for the shortest possible time. Care should be taken to avoid bacterial contamination and cross-contamination with other allergens. They should be discarded after the expiry date.
INTERPRETATION OF SPT RESULTS
For positive control (histamine), a wheal diameter of ≥3 mm (at least 3 mm greater than negative control) is taken as positive.[8] A wheal diameter of <3 mm for positive control might be seen if the patient is taking drugs with antihistamine activity or has non-reactive skin. In this case, it is not possible to go ahead with the SPT.
A wheal >3 mm for negative control (normal saline) indicates a highly reactogenic skin (e.g., dermatographism), making SPT invalid.[24]
In case of individual allergens, a wheal diameter of ≥3 mm (at least 3 mm greater than negative control) is taken as a positive test [Figure 2]. The larger the wheal, the greater the likely hood of an allergic reaction when exposed to the allergen. However, a larger size does not necessarily correspond to more severe reaction.
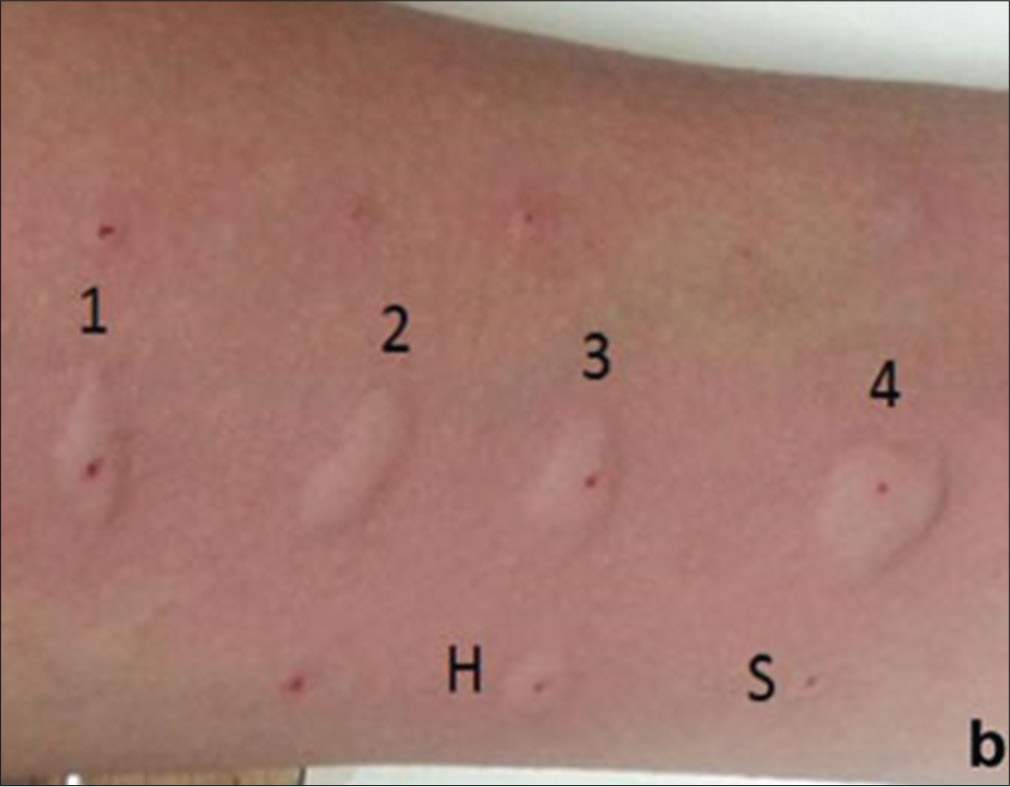
- Picture showing wheal and flare reaction after a skin prick testing using histamine (h), saline (s), and various allergens (marked 1–4). Reproduced with permission from Gupta N, Indian pediatrics 2019.
False-positive reactions can occur in certain skin conditions (e.g., dermatographism), naturally occurring histamine in some allergen extracts, non-standard reagents, and cross-reactivity with allergens with homologous proteins.[8]
False-negative tests can occur in patients taking drugs with antihistamine activity, recent history of anaphylaxis (<4 weeks), non-standard reagents, improper technique, and UV light exposure.[8]
ADVANTAGES OF SPT
When performed and interpreted correctly, SPT is highly sensitive for IgE-mediated allergies.[25]
They are relatively cheap and easy to interpret. This is particularly important in a country like India.
When right technique is used, SPT is minimally invasive and should cause very little discomfort.
The results are immediately available to the family and necessary counseling can be done at the same visit. This visible reaction may act as a motivation to the family for allergen avoidance measures and appropriate treatment.
Unlike in vitro tests, the results may not affected by conditions associated with high total IgE levels.[8]
Limitations of SPT
SPT results can be variable and depend on the quality of the reagent, the technique used and proper interpretation of results.[6]
The availability of standard reagents can be a challenge in our country. This can affect the quality of the test.
It needs some normal area of skin to test and cannot be done in patients where extensive area of skin is involved.
SPT cannot be done where the patient cannot stop taking drugs with antihistamine activity that can interfere with the results.
The testing requires patient cooperation and may be slightly difficult in very young infants.
Because of the possibility of systemic allergic reactions, they should be performed only in settings where severe allergic reactions can be managed.
SIDE EFFECTS OF SPT
Some patients experience mild discomfort and itching at the site of SPT. This is usually transient and self-limiting. Rarely, delayed local, painful swelling may occur due to IgE-mediated delayed reaction.
Non-allergic reactions such as infection, headache, syncope, and malaise are very rare.
Although very rare, systemic allergic reactions (including anaphylaxis) are known to occur following SPT.[26] Valyasevi et al. studied the incidence of systemic reactions in 497,656 skin tests and found 33 systemic reactions per 100,000 skin tests.[26] However, the reactions were mild and recovered within 1 h.
The incidence of systemic reactions was higher when testing was done for latex, antibiotics, and food allergy (especially when raw foods are used) and intradermal method of testing was used. Patient factors that were associated with increased risk of systemic reactions include young infants, pregnant women, uncontrolled asthma, previous history of anaphylaxis,[27] patients using ACE inhibitors and beta-blockers (diminished response to adrenaline treatment), and elevated baseline tryptase levels. Clinicians should exercise extra caution in the above circumstances.
CONCLUSION
SPT is a safe, reliable, and sensitive test. It is considered the gold standard for diagnosis of aeroallergens and is considered superior to sIgE. However, it is inferior to oral food challenge in diagnosis of food allergies.
It is inexpensive and the results are immediately available; definite advantages in a country like ours.
Once diagnosis of allergy is confirmed by SPT, targeted allergen avoidance measures can be advised.
An appropriate allergy management plan including recognition and management of severe allergic reactions can be given to the family. Immunotherapy can be advised where appropriate.
The performance and interpretation of the SPTs should be done by a person who is trained in diagnosis and management of allergy in children.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Allergies in India: An analysis of 3389 patients attending an allergy clinic in Mumbai, India. J Indian Med Assoc. 2008;106:220-2.
- [Google Scholar]
- A comparative study of skin prick test versus serum-specific IgE measurement in Indian patients with bronchial asthma and allergic rhinitis. Indian J Chest Dis Allied Sci. 2015;57:81-5.
- [Google Scholar]
- Advances in food allergy diagnosis. Curr Pediatr Rev. 2018;14:139-49.
- [CrossRef] [PubMed] [Google Scholar]
- Role and interpretation of total serum IgE measurements in the diagnosis of allergic airway disease in adults. Allergy. 2003;58:905-11.
- [CrossRef] [PubMed] [Google Scholar]
- Allergy diagnosis: Pros and cons of different tests, indications and limitations. Breathe. 2007;3:345-9.
- [CrossRef] [Google Scholar]
- Comparison of the performance of skin prick, immuneCAP, and ISAC tests in the diagnosis of patients with allergy. Int Arch Allergy Immunol. 2017;172:215-23.
- [CrossRef] [PubMed] [Google Scholar]
- Skin Prick Testing for the Diagnosis of Allergic Disease-a Manual for Practitioners Australia: Australasian Society of Clinical Immunology and Allergy; 2016.
- [Google Scholar]
- The skin prick test-European standards. Clin Transl Allergy. 2013;3:3.
- [CrossRef] [PubMed] [Google Scholar]
- Common environmental allergens causing respiratory allergy in India. Indian J Pediatr. 2002;69:245.
- [CrossRef] [PubMed] [Google Scholar]
- Comparative study of food allergies in children from China, India, and Russia: The EuroPrevall-INCO surveys. J Allergy Clin Immunol. 2020;8:1349-58.
- [CrossRef] [PubMed] [Google Scholar]
- Allergy diagnostic testing: An updated practice parameter. Ann Allergy Asthma Immunol. 2008;100:S1-48.
- [CrossRef] [Google Scholar]
- Long-term oral corticosteroid therapy does not alter the results of immediate-type allergy skin prick tests. J Allergy Clin Immunol. 1996;98:522-7.
- [CrossRef] [Google Scholar]
- Prolonged treatment with topical glucocorticoids results in an inhibition of the allergen-induced weal-and-flare response and a reduction in skin mast cell numbers and histamine content. Clin Exp Allergy. 1989;19:19-25.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of topical steroids on skin prick test: A randomized controlled trial. Dermatol Ther. 2018;8:285-90.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of topical corticosteroid application frequency on histamine-induced wheals. Int J Dermatol. 2005;44:425-7.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of topical pimecrolimus on epicutaneous skin testing. J Allergy Clin Immunol. 2004;114:695.
- [CrossRef] [PubMed] [Google Scholar]
- Suppressive effects of topical mometasone furoate and tacrolimus on skin prick testing in children. Pediatr Dermatol. 2008;25:269-70.
- [CrossRef] [PubMed] [Google Scholar]
- Investigation on the effects of topical therapy with 0.1% tacrolimus ointment (Protopic®) on intradermal skin test reactivity in atopic dogs. Vet Dermatol. 2004;15:218-24.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of distance between sites and region of the body on results of skin prick tests. J Allergy Clin Immunol. 1996;97:596-601.
- [CrossRef] [Google Scholar]
- The relationship between horse dandruff and horse serum antigens in asthma. Clin Sci. 1950;9:127.
- [Google Scholar]
- Measurement and interpretation of skin prick test results. Clin Transl Allergy. 2015;6:1-5.
- [CrossRef] [PubMed] [Google Scholar]
- Allergen immunotherapy In: Allergic Diseases. United States: Humana Press; 2007. p. :429-44.
- [CrossRef] [Google Scholar]
- Skin testing versus in vitro testing in the evaluation of aeroallergy: The great debate. Am J Rhinol Allergy. 2015;29:362-4.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic reactions to allergy skin tests. Ann Allergy Asthma Immunol. 1999;83:132-6.
- [CrossRef] [Google Scholar]
- Systemic reactions from skin testing: Literature review. J Investig Allergol Clin Immunol. 2006;16:75.
- [Google Scholar]






